A) energy that flows between objects with different temperatures.
B) movement of ions or electrons.
C) stored energy that could do work but is not doing so.
D) energy that moves in waves.
E) the form of energy that actually does work.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When ionic compounds dissolve in water, their ions
A) dissociate or separate from one another.
B) get lost in the solvent.
C) cling tightly together.
D) lose their charge.
E) settle to the bottom of the container.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The building blocks of proteins are
A) eicosanoids.
B) amino acids.
C) triglycerides.
D) phospholipids.
E) monosaccharides.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is mismatched?
A) triglyceride--fat
B) eicosanoid--prostaglandin
C) ribose--RNA
D) enzyme--protein
E) cholesterol--nucleic acid
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following five terms. Which term does not belong with the other four terms?
A) glucose
B) lactose
C) disaccharide
D) sucrose
E) maltose
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Adjacent amino acids in a polypeptide chain are held together by
A) hydrogen bonds.
B) high energy bonds.
C) peptide bonds.
D) Van der Waals bonds.
E) ionic bonds.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What protein structure results from folding or coiling of a polypeptide chain caused by hydrogen bonds between amino acids?
A) secondary structure
B) tertiary structure
C) quaternary structure
D) primary structure
E) peptide structure
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
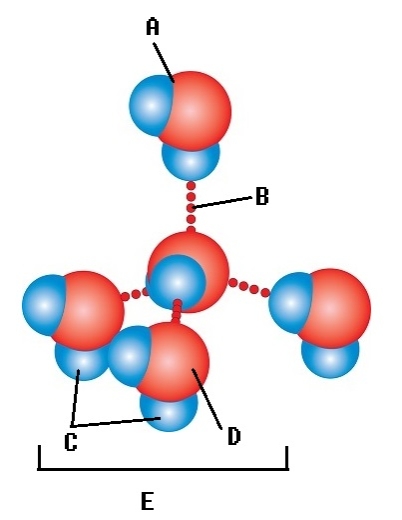 -Water accounts for 50% of the weight of a young adult female and 60% of a young adult male. What kind of bond is found at "B"?
-Water accounts for 50% of the weight of a young adult female and 60% of a young adult male. What kind of bond is found at "B"?
A) oxygen atom
B) polar covalent bond
C) water molecule
D) hydrogen atom
E) hydrogen bond
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How does a buffer help a solution maintain pH?
A) A buffer releases acid to maintain proper pH.
B) A buffer forms both cations and anions to counteract acids.
C) A buffer can act like a base if pH is acidic, and it can act like an acid if pH is basic.
D) A buffer release base to neutralize acid.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The energy stored in ATP is a form of ________ energy.
A) mechanical
B) heat
C) electrical
D) chemical
E) kinetic
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
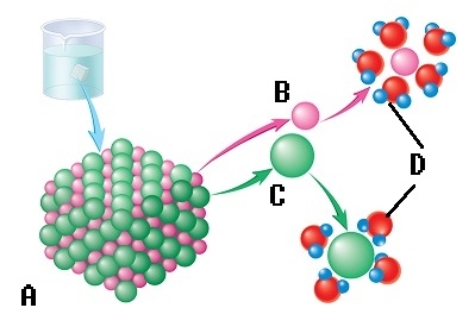 -The sodium chloride molecule breaks apart in water. What does "A" represent?
-The sodium chloride molecule breaks apart in water. What does "A" represent?
A) chloride ion
B) dissociation
C) water molecule
D) sodium ion
E) salt crystal
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Chemical energy
A) moves matter.
B) results from the position or movement of objects.
C) is a form of potential energy within chemical bonds.
D) comes from the sun.
E) is not important in physiological processes.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The molecule used most frequently by cells as a fuel belongs to which of the following groups?
A) carbohydrates
B) nucleic acids
C) prostaglandins
D) phospholipids
E) steroids
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Normal pH range for blood is 7.35 to 7.45. If blood pH falls below 7.35,
A) the number of red blood cells decreases.
B) the blood becomes saltier.
C) an imbalance called acidosis results.
D) an imbalance called alkalosis results.
E) nothing happens as this is an acceptable deviation.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hyperventilation causes the loss of large amounts of carbon dioxide from the body, decreasing the amount of hydrogen ions in solution. As a result,
A) the pH of body fluids will rise.
B) the pH of body fluids will fall.
C) the pH of body fluids will become neutral.
D) the pH of body fluids will not be affected.
E) None of these choices is correct.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A base is a proton
A) donor.
B) converter.
C) acceptor.
D) creator.
E) Both acceptor and creator.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydrogen bonds are important for all of the following except:
A) producing surface tension in water
B) helping hold DNA strands together
C) helping atoms give up or receive electrons
D) helping hold a protein structure together
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a synthesis reaction?
A) Two amino acids are bonded together to form a dipeptide.
B) Sodium chloride is dissolved in water.
C) ATP is converted to ADP.
D) Sucrose is chemically separated to form one molecule of glucose and one molecule of fructose.
E) Several dipeptide chains are formed from digestion of a long polypeptide chain.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If the products of a chemical reaction contain less potential energy than the reactants,
A) energy has been stored in the molecular bonds of the product.
B) energy has been released by the breaking of molecular bonds.
C) the reaction will be reversible without additional energy input.
D) a synthesis reaction is likely to have occurred.
E) All of these choices are correct.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Cations and anions that dissociate in water are sometimes called
A) molecules
B) nonelectrolytes and solutes.
C) electrolytes, because they can conduct an electrical current.
D) nonelectrolytes, because they do not conduct an electrical current.
E) molecules and electrolytes.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 121 - 140 of 168
Related Exams