A) C
B) P
C) As
D) Ga
E) Sb
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of these elements has seawater as its commercial source
A) Mg
B) Al
C) Ca
D) Sn
E) Cd
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Cryolite, Na3AlF6, is mixed with alumina prior to electrolysis in the production of Al because it acts a solute.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A naturally occurring substance with a range of chemical composition is
A) an element.
B) a mineral.
C) gangue.
D) an ore.
E) an amalgam.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the Mond process, nickel is purified by
A) dissolving the crude ore in sulfuric acid, and precipitating the nickel(II) ion as the sulfate salt.
B) dissolving the crude ore in a lye (NaOH) solution, and precipitating the nickel(II) ion as the hydroxide salt.
C) heating the crude ore to its melting point and electrolytically reducing the nickel(II) ions to form the metal.
D) passing sodium gas over the crude ore at high temperatures, chemically reducing the nickel(II) to nickel metal.
E) passing carbon monoxide over the impure metal at high temperature, forming the volatile Ni(CO) 4 which is removed as a gas.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these elements when doped into silicon would give an n-type semiconductor
A) C
B) Ga
C) P
D) Ge
E) B
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following diagrams represents a semiconductor 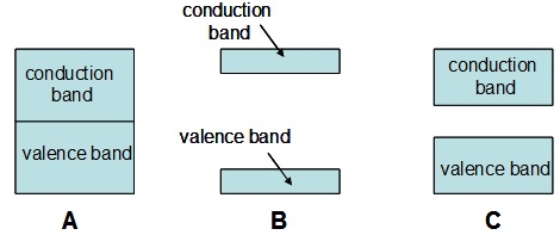
A) A
B) B
C) C
E) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following reaction is used to produce titanium metal at high temperature.TiCl4(g) + 2Mg(l) Ti(s) + 2MgCl2(l) Which element is oxidized and which is reduced
A) Both titanium and magnesium are oxidized
B) Titanium is reduced, magnesium is oxidized
C) Magnesium is reduced, titanium is oxidized
D) Neither titanium or magnesium is oxidized
E) None of the above
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the total number of moles (n) of electrons exchanged between the oxidizing agent and the reducing agent in the reaction below to obtain vanadium metal V2O5(s) + 5Ca(l) 2V(l) + 5CaO(s)
A) 1
B) 2
C) 5
D) 10
E) 0
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to the band theory, which of these statements provide(s) an explanation for the high electrical conductivity of metals I. a partly filled conduction band II. a valence band overlapping an empty conduction band III. a filled valence band IV. a large gap between the valence band and the conduction band
A) I and II
B) I and III
C) III
D) III and IV
E) IV
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Write the chemical formula of epsomite (sold in pharmacies as Epsom salts) .
A) CaSO4 7H2O
B) MgSO4 7H2O
C) Na2SO4 7H2O
D) FeSO4 7H2O
E) None of the above
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What effect does increasing temperature have on the conductivities of semiconductors
A) increases
B) decreases
C) no change
D) cannot be predicted
F) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Bauxite (Al2O3 2H2O) ore is the principal commercial source of aluminum metal.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of these metals would normally be obtained by chemical reduction
A) aluminum
B) calcium
C) lithium
D) sodium
E) vanadium
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The equilibrium 3Fe(s) + C(s) Fe3C(s) is established in a solid solution. For such a solution, one can write an equilibrium constant expression in the usual way except that the concentrations that refer to solids in the solid solution are included. Determine the equilibrium constant for the formation of cementite from iron and carbon at 680 C. (Given: for this reaction at 25 C, H = 21 kJ/mol and S = 20.4 J/mol.K)
A) 0.75
B) 0.33
C) 3.1
D) 0.82
E) 1.2
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The principal reducing agent in a blast furnace is
A) CaO(s)
B) CaSiO3(l)
C) CO(g)
D) O2(g)
E) CO2(g)
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The naturally occurring form of a metal that is concentrated enough to allow economical recovery of the metal is known as
A) an element.
B) a mineral.
C) gangue.
D) an ore.
E) an amalgam.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Volatile impurities are removed from ores by means of
A) roasting.
B) amalgamation.
C) electrolysis.
D) flotation.
E) zone refining.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Aluminum hydroxide, Al(OH) 3, is
A) an acid.
B) an amphoteric hydroxide.
C) a base.
D) an explosive hydroxide.
E) used to make amalgams.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Mercury, magnesium, and zinc have low enough boiling points that they can be purified by distillation.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 63
Related Exams